Unieke apparaatidentificatie - Markering van medische hulpmiddelen
Vanaf 26 mei 2021 moeten fabrikanten, distributeurs, importeurs en door de EU gemachtigde vertegenwoordigers van medische hulpmiddelen of hun verpakking op grond van de EU-verordening betreffende medische hulpmiddelen (MDR) 2017/745 duidelijk worden gemerkt. In de medische technologie zullen medische hulpmiddelen daarom volledig traceerbaar zijn van fabrikant tot gebruiker en de door de EU vereiste patiëntveiligheid garanderen.

Unieke apparaatidentificatie (UDI)
Vanaf 26 mei 2021 moeten fabrikanten, distributeurs, importeurs en gemachtigde vertegenwoordigers in de EU van medische hulpmiddelen of hun verpakking een duidelijk etiket dragen op grond van de EU-verordening betreffende medische hulpmiddelen (MDR) 2017/745. In de medische technologie zullen medische hulpmiddelen zo volledig traceerbaar zijn van fabrikant tot gebruiker en de door de EU vereiste patiëntveiligheid garanderen.
De normen en voorschriften die zijn aangenomen in overeenstemming met de verordening betreffende medische hulpmiddelen zijn even bindend voor alle belanghebbenden. De voor het medische hulpmiddel vereiste productidentificatie wordt toegewezen door een van de vier toewijzende instanties - GS1, HIBCC, ICCBBA en het Informatiecentrum voor farmaceutische specialiteiten.
Vanaf mei 2022 worden alle medische hulpmiddelen met hun stamgegevens en de unieke hulpmiddelidentificatie (UDI) opgeslagen in de EU-brede database EUDAMED.
De productidentificatie bestaat uit twee componenten
- Device Identifier (DI): een statische code met ongeveer 20 gegevens voor identificatie van fabrikant en product
- Production Identifier (PI): variabele gegevens gebruikt voor traceerbaarheid, zoals batchnummer, vervaldatum of serienummer
Uitvoeringstermijnen
Etiketteringsverplichting voor risicoklassen

Waar moet de UDI-markering worden aangebracht?
Directe markering met inkt, laser of etiket
De productidentificatie moet rechtstreeks op het product, op de primaire verpakking of op alle hogere verpakkingsniveaus worden aangebracht. Volgens de Medical Device Regulation moet de directe markering worden aangebracht met inkt, laser of etiket. Bovendien moet de code gemakkelijk toegankelijk en leesbaar zijn, zowel in het magazijn als wanneer het product in gebruik is - en leesbaar blijven gedurende de volledige levensduur van het product.
UDI podcast van het Johner Instituut
Luister naar de podcast van het Johner Institute over het onderwerp UDI met Prof. D. Christian Johner en de heer Wilfried Weigelt.
Markering is altijd in platte tekst en als een machineleesbare code. Afhankelijk van het oppervlak dat beschikbaar is voor rechtstreekse markering, kunnen 1D-barcodes of 2D-matrix codes op de medische hulpmiddelen worden aangebracht.
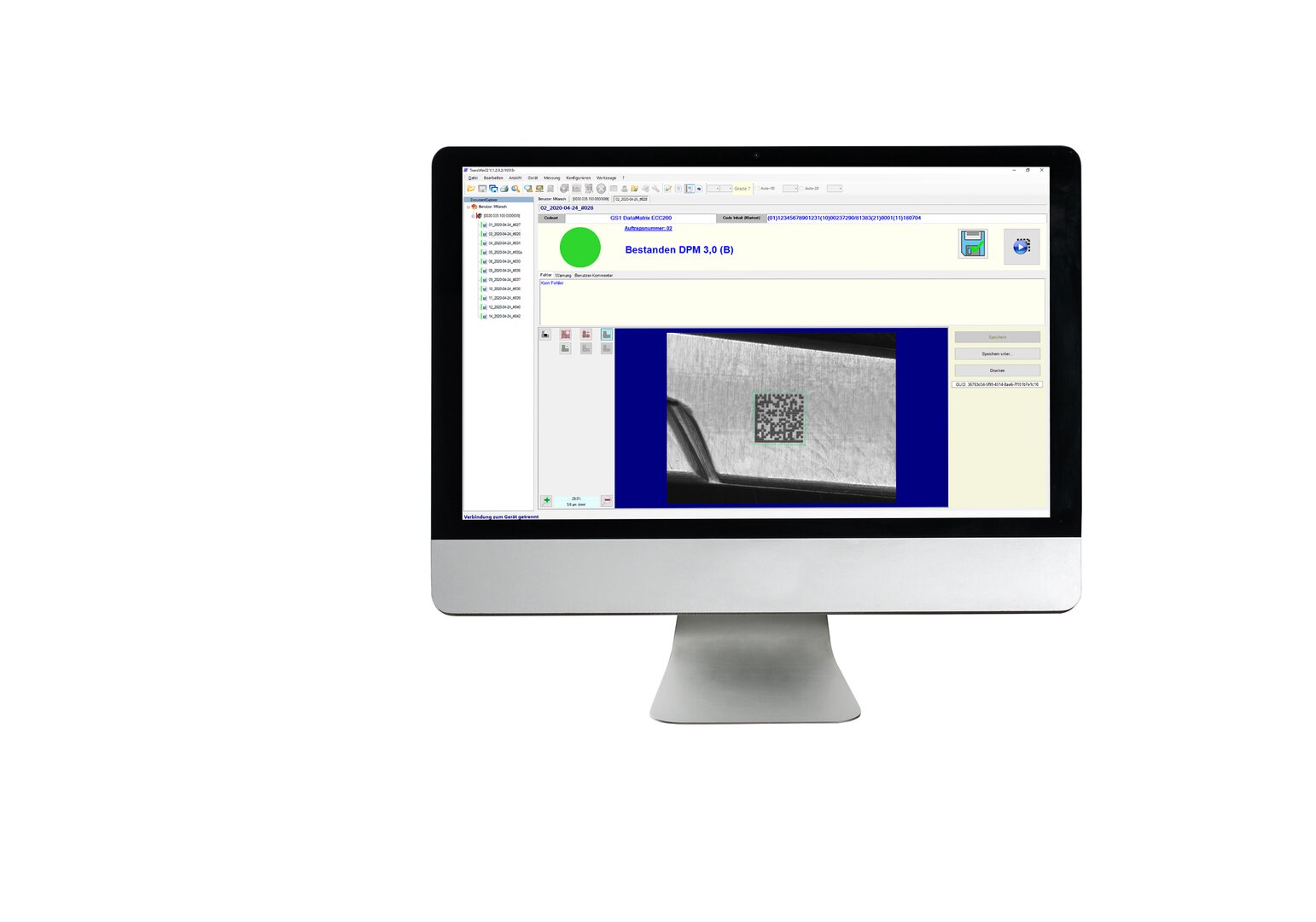
Afhankelijk van het medische hulpmiddel en de eigenschappen ervan, is er een zeer breed scala aan mogelijke varianten van markering. De verificatiesystemen voor codes van REA VERIFIER worden gebruikt om ervoor te zorgen dat de codes foutloos worden gelezen met een hoog eerste leespercentage en dat wordt voldaan aan de normen van de verordening inzake medische hulpmiddelen.
Hoge resolutie inkjetprinters gebaseerd op HP cartridges
REA JET HR codeer- en markeersystemen op basis van HP cartridges zijn ideaal voor 2D-codes met hoge resolutie, merklogo's of andere markeringen van hoge kwaliteit - op hoge snelheden. Producten en verpakkingen kunnen worden gemarkeerd met een printhoogte van 12,7 mm per printkop - meerdere printkoppen kunnen in cascade worden geschakeld voor grotere printhoogten. Hoge eerste leespercentages zijn daarom geen probleem op het verkooppunt.

REA LASER CL
REA LASER CL lasersystemen markeren verpakkingen van organische materialen permanent, zeer nauwkeurig en individueel. De lasersystemen zijn verbruiksvrij. Ook hier zijn hoge eerste leespercentages gegarandeerd.

REA LASER FL
REA LASER FL lasersystemen markeren contactloos, precies en individueel kunststof verpakkingen. Fiber laser markering is permanent en ideaal voor hardere metalen zoals roestvrij staal en titanium en voor kunststoffen en dunne folies. De lasersystemen zijn verbruiksvrij. Ook hier zijn hoge eerste leespercentages op het verkooppunt gegarandeerd.

Etiketteertechnologie
Etiketteeroplossingen zijn ideaal wanneer direct markeren met inkjet printers of lasermarkeersystemen geen optie is.
REA LABEL biedt hiervoor geschikte oplossingen, van eenvoudige tafelmodel etikettenprinters tot complexe etiketteersystemen voor alle toepassingen.

Codecontrolesystemen
Verificatiesystemen van REA VERIFIER worden gebruikt om te garanderen dat markeringen en codes foutloos zijn en voldoen aan internationale normen. Op deze manier krijgt elke markering een acceptatietestrapport.
REA biedt een compleet productassortiment en code-expertise voor de uitvoering van deze taak. Neem contact met ons op.

REA is uw partner
Advies en training
- REA adviseert en traint u in UDI-aangelegenheden
- Inschakelen van deskundige partners voor gegevensbeheer
- Geschiktheid en gebruik van codeer- en markeertechnologieën
- Geschiktheid en gebruik van verificatiesystemen voor naleving van specificaties voor codekwaliteit